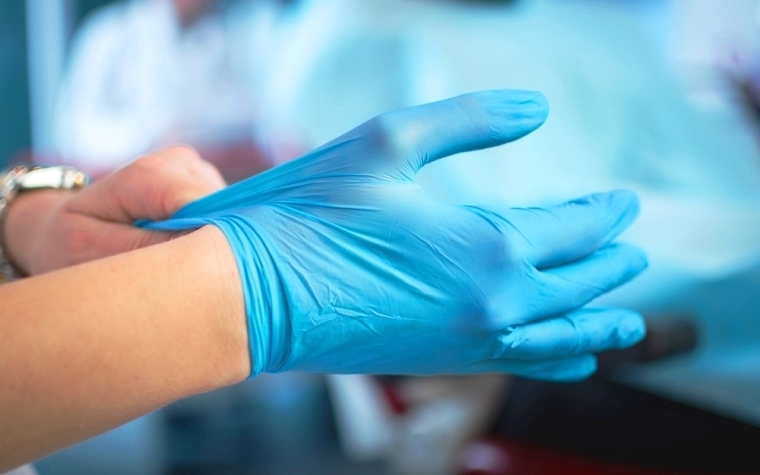
The U.S. Food and Drug Administration recently reminded medical professionals nationwide that effective this week, the majority of powdered medical gloves will be banned from use following its regulation passed in December 2016.
The decision, first proposed in March 2016, applies to surgeon’s gloves and patient examination gloves that have been powdered, along with the absorbable powder used to lubricate surgeon’s gloves.
The agency listed risks including inflammation and respiratory allergic reactions. Although use had decreased, the FDA stated that the gloves still “pose an unreasonable and substantial risk of illness or injury to … individuals who are exposed to them, which cannot be corrected through new or updated labeling. Consequently, FDA is banning these devices.”
The sanction was enacted under the Department of Health and Human Services and states that the treated gloves present too high of a health risk for continued usage.
“This ban is about protecting patients and health care professionals from a danger they might not even be aware of,” Dr. Jeffrey Shuren, director of the FDA’s Center for Devices and Radiological Health, said. “We take bans very seriously and only take this action when we feel it’s necessary to protect the public health.”
The FDA does not expect the ban to cause any shortage of glove supplies as numerous non-powdered products are available.