
05/03/23
First-Quarter 2023 Results:
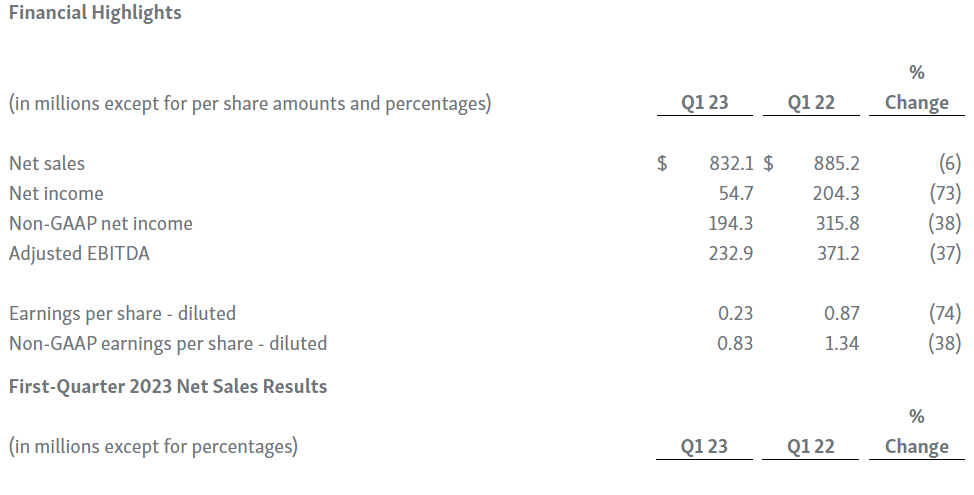
-- Net Sales of $832.1 Million --
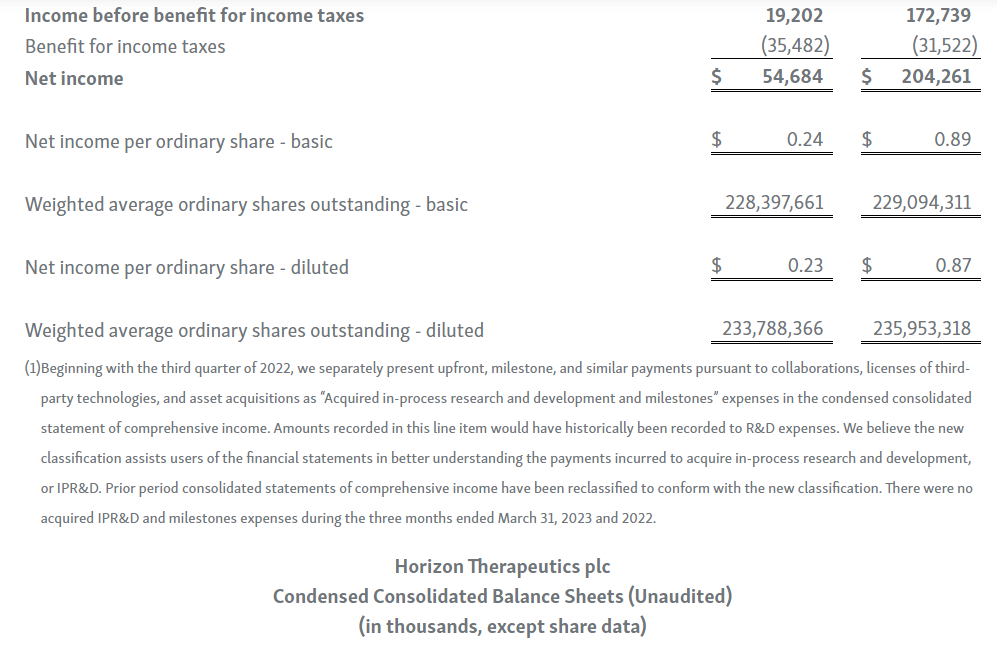
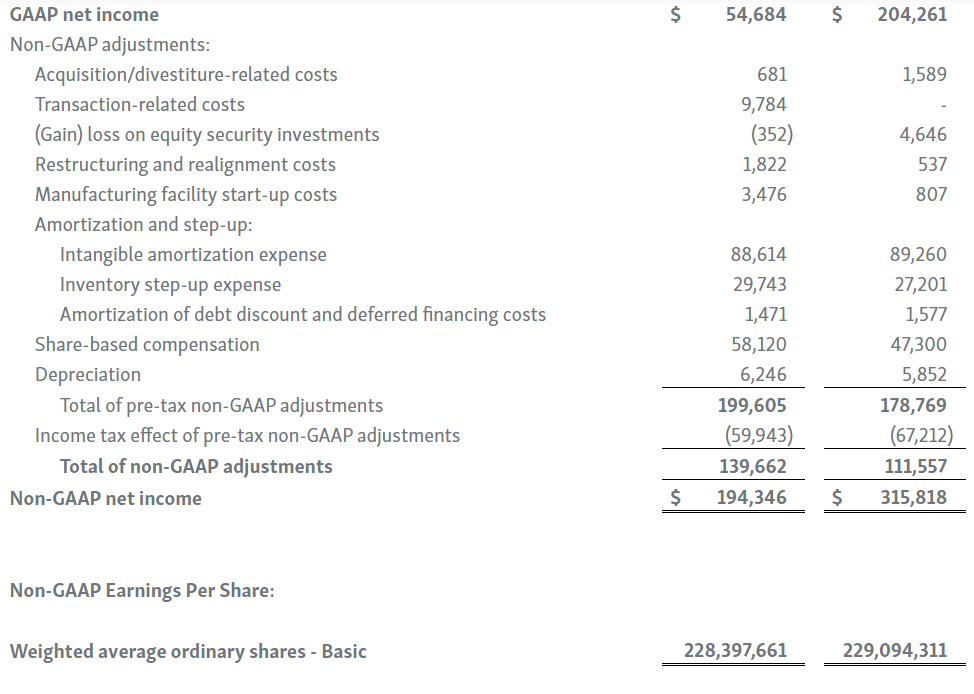
-- GAAP Net Income of $54.7 Million; Adjusted EBITDA of $232.9 Million --
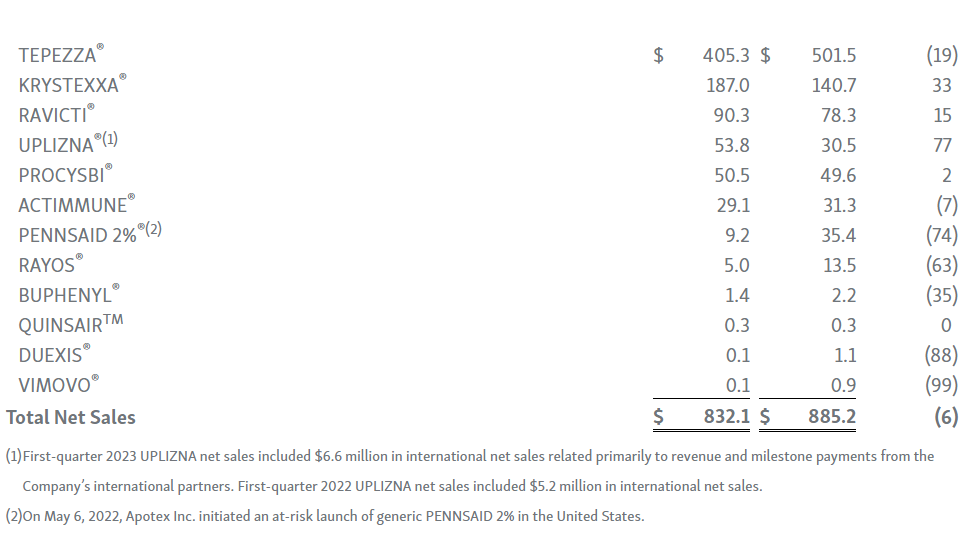
-- TEPEZZA® (teprotumumab-trbw) Net Sales of $405.3 Million --
-- KRYSTEXXA® (pegloticase injection) Net Sales of $187.0 Million --
-- UPLIZNA® (inebilizumab-cdon) Net Sales of $53.8 Million --
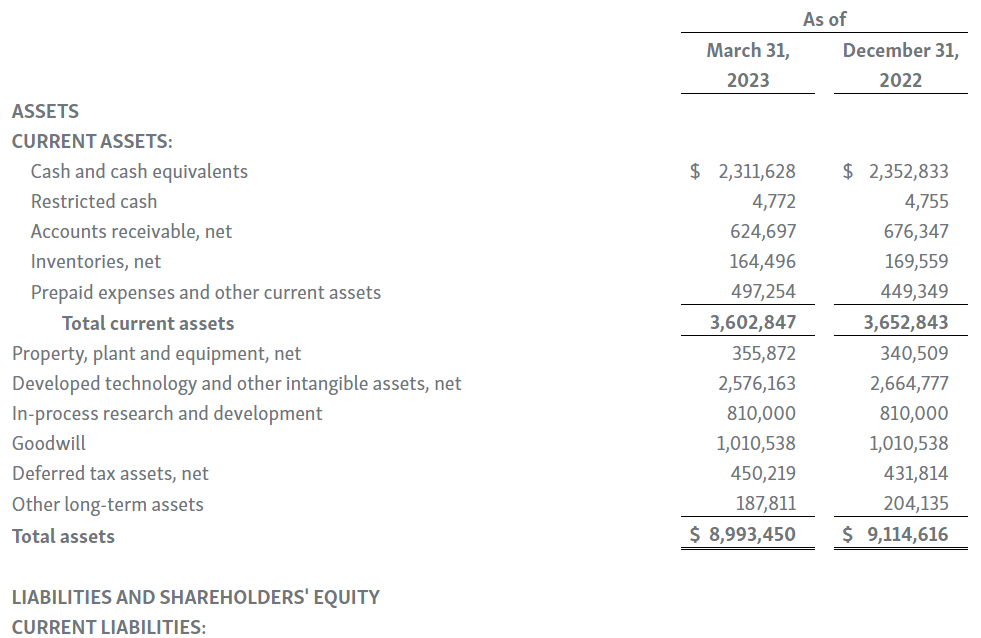
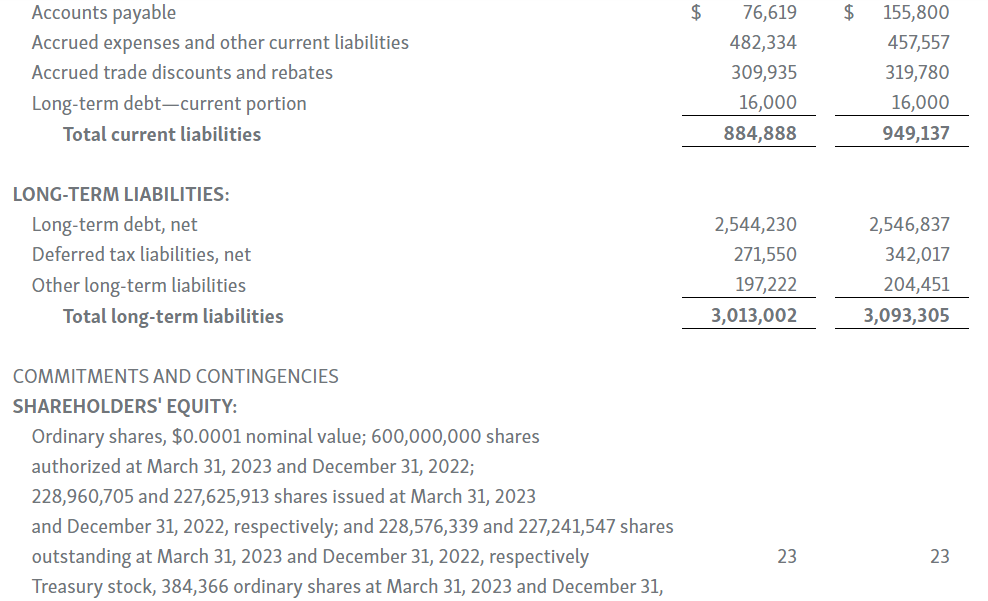
-- Cash Position of $2.31 Billion as of March 31, 2023 --
First-Quarter and Recent Company Highlights:
-- Announced Positive Topline Data from TEPEZZA Phase 4 Clinical Trial in Patients with Chronic/Low Clinical Activity Score (CAS) Thyroid Eye Disease (TED) --
-- Obtained U.S. FDA Approval for Updated TEPEZZA Indication to Specify Treatment of TED Patients Regardless of Disease Activity or Duration --
-- Announced Positive Topline Data from the Second Population in Dazodalibep Sjögren’s Syndrome Phase 2 Clinical Trial; First and Only Phase 2 Sjögren’s Syndrome Trial to Meet the Primary Endpoint in Both Patient Populations; Phase 3 Clinical Program Expected to Initiate in 2023 --
-- Announced Initiation of TEPEZZA Phase 3 Trial in Japan in Chronic/Low CAS TED and Two Daxdilimab Phase 2 Trials in Discoid Lupus Erythematosus and Lupus Nephritis --
-- Continue to Expect Amgen Transaction to Close in 1H23 --
-- Named One of Fortune’s 100 Best Companies to Work For® and Ranked as Top Biotechnology/Pharmaceutical Company, Both for Third Consecutive Year --
DUBLIN--(BUSINESS WIRE)--May 3, 2023-- Horizon Therapeutics plc (Nasdaq: HZNP) today announced first-quarter 2023 financial results.
“We generated strong first-quarter performance, with double-digit growth for KRYSTEXXA and UPLIZNA, and saw positive trends in TEPEZZA's leading indicators as we exited the first quarter,” said Tim Walbert, chairman, president and chief executive officer, Horizon. “Importantly, we announced positive topline results from our TEPEZZA clinical trial in low CAS and long-duration TED patients, as well as received FDA approval for an update to the TEPEZZA indication that reinforces the potential benefit of TEPEZZA, regardless of disease activity or duration. These important events will help us to ease the access burden so all eligible patients can benefit from TEPEZZA. I am extremely proud of what we have accomplished in a few short months and believe we are well positioned as we prepare to become part of Amgen.”
Key Growth Drivers
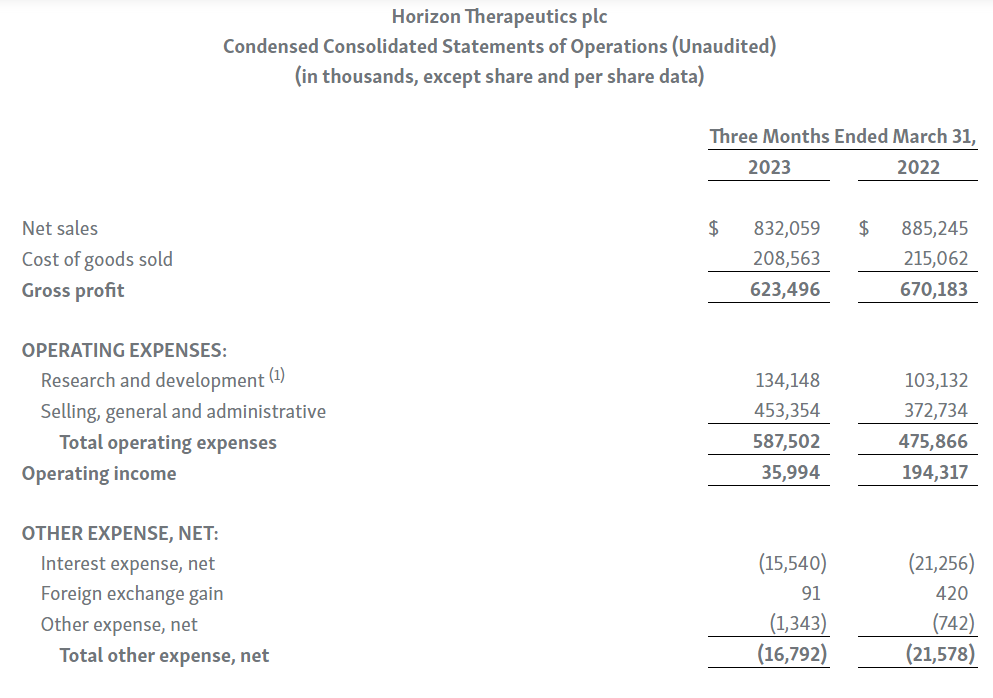
TEPEZZA: TEPEZZA net sales in the first quarter were $405 million, representing an 18 percent sequential decline compared to the fourth quarter of 2022. Net sales were impacted by seasonality, which the Company typically sees with its infused medicines in the first quarter each year. In addition, the TEPEZZA field-force expansion initiated late in 2022 has not yet substantially impacted net sales. More recently, the Company has seen the expansion drive positive momentum in the business, including increases in new prescribers, patient enrollment forms and patient starts. As new prescribers and patients work through the reimbursement process, the Company expects a more meaningful impact to net sales later in the year.
In April 2023, the Company announced positive topline results from its TEPEZZA Phase 4 clinical trial in patients with low CAS and long-duration TED and received FDA approval for an update to the indication for TEPEZZA that supports its potential benefit in TED, regardless of disease activity or duration. This reinforces the importance of unrestricted access for eligible patients across the full spectrum of TED and creates an opportunity to ease the access burden for patients and physicians and to decrease time to therapy for patients who may benefit from TEPEZZA. The Company plans to present data from the Phase 4 trial at a future medical congress and publish the data in a peer-reviewed medical journal to help educate key stakeholders, including physicians, patients and payors.
KRYSTEXXA: KRYSTEXXA first-quarter net sales were $187 million, representing a year-over-year increase of 33%. Sequentially, net sales declined by 13 percent compared to fourth-quarter 2022 due to seasonality. Strong performance in the first quarter was driven by the continued momentum in both the rheumatology and nephrology market segments, including the adoption of KRYSTEXXA with immunomodulation as the new standard of care following FDA approval in July 2022 for an expanded label to include KRYSTEXXA with methotrexate. The Company’s efforts to educate physicians and key stakeholders continues to lead to strong patient growth from both new and existing prescribers across both market segments.
UPLIZNA: UPLIZNA first-quarter 2023 net sales were $53.8 million, representing a year-over-year increase of 77%, driven by continued strong execution. Net sales in the U.S. were $47.2 million, an increase of 87%, and were driven by strong and consistent growth in new prescribers and new patient starts. The Company continues to advance its global expansion strategy with multiple planned international launches in 2023.
Conference Call
In light of the announced agreement to be acquired by Amgen Inc. and applicable securities laws, the Company will not be hosting a conference call to discuss its financial results. This earnings press release and the related Quarterly Report on Form 10-Q for the quarter ended March 31, 2023 are publicly available in the Investor Relations section of the Company’s website at https://ir.horizontherapeutics.com.
Horizon is a global biotechnology company focused on the discovery, development and commercialization of medicines that address critical needs for people impacted by rare, autoimmune and severe inflammatory diseases. Our pipeline is purposeful: we apply scientific expertise and courage to bring clinically meaningful therapies to patients. We believe science and compassion must work together to transform lives. For more information on how we go to incredible lengths to impact lives, visit www.horizontherapeutics.com and follow us on Twitter, LinkedIn, Instagram and Facebook.
Note Regarding Use of Non-GAAP Financial Measures
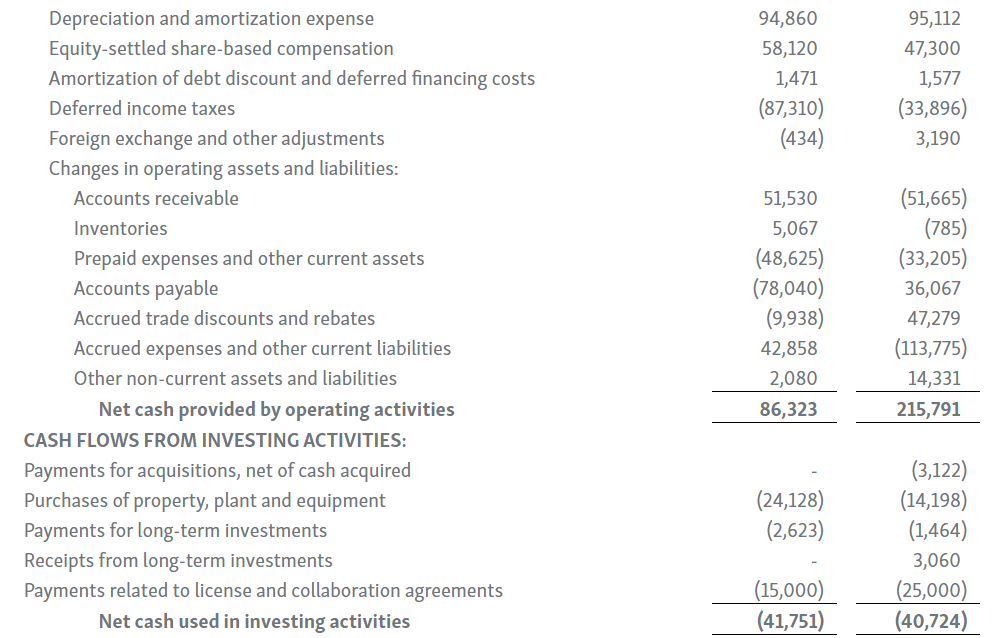
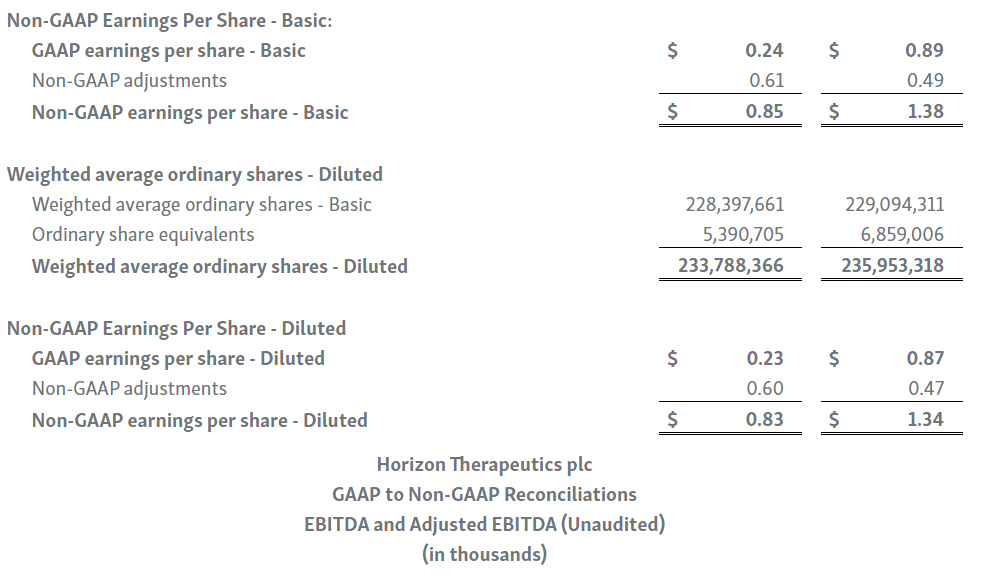
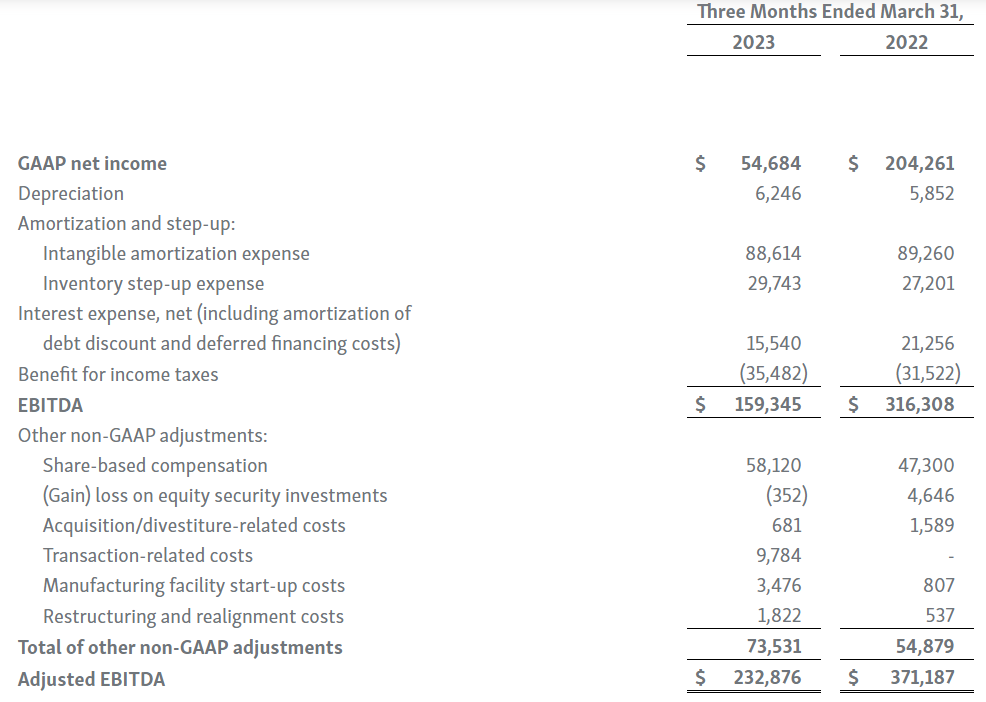
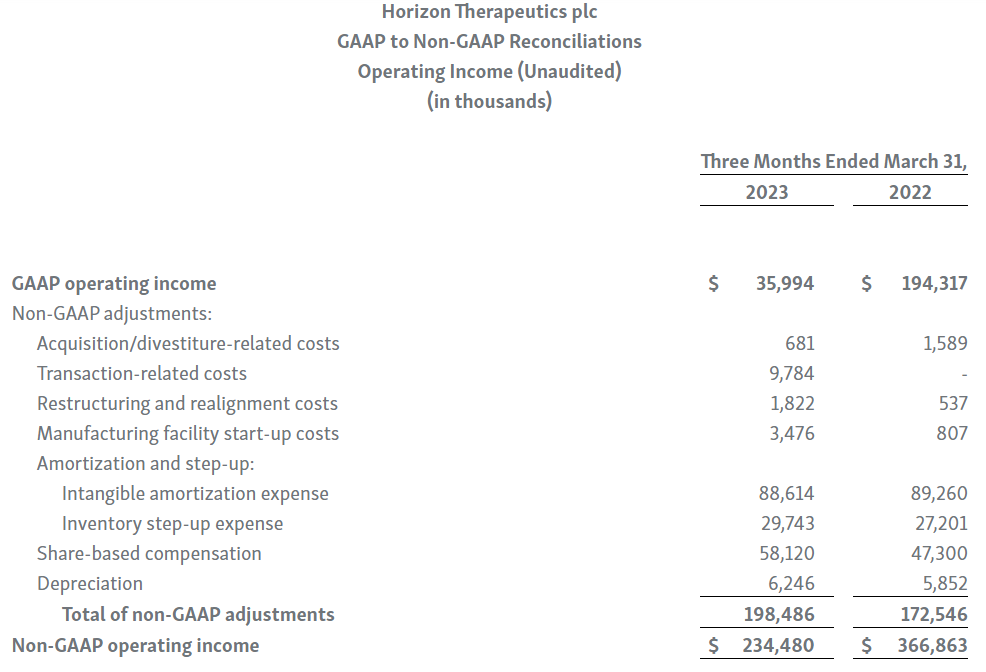
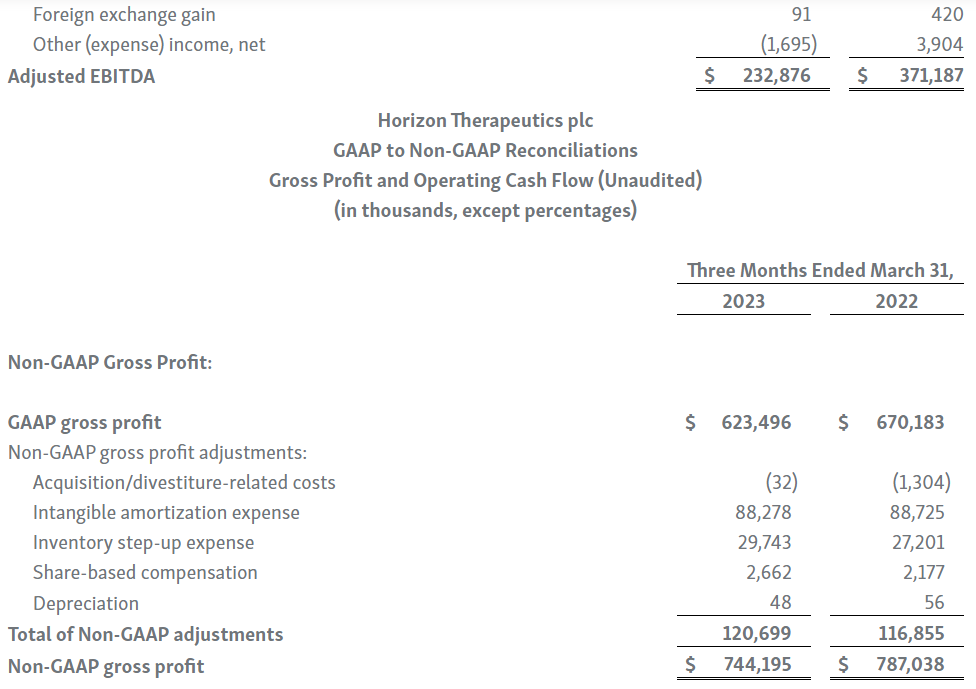
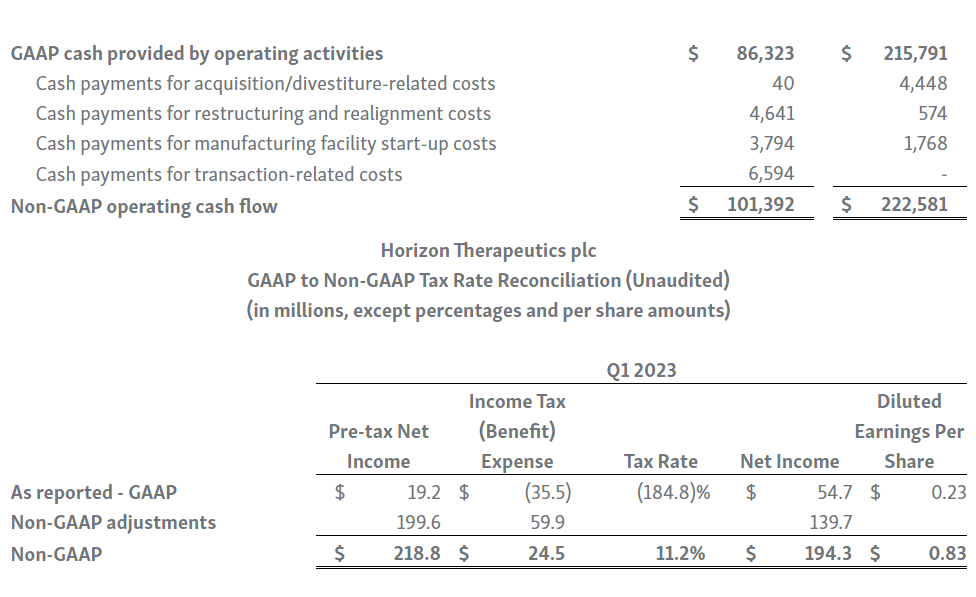
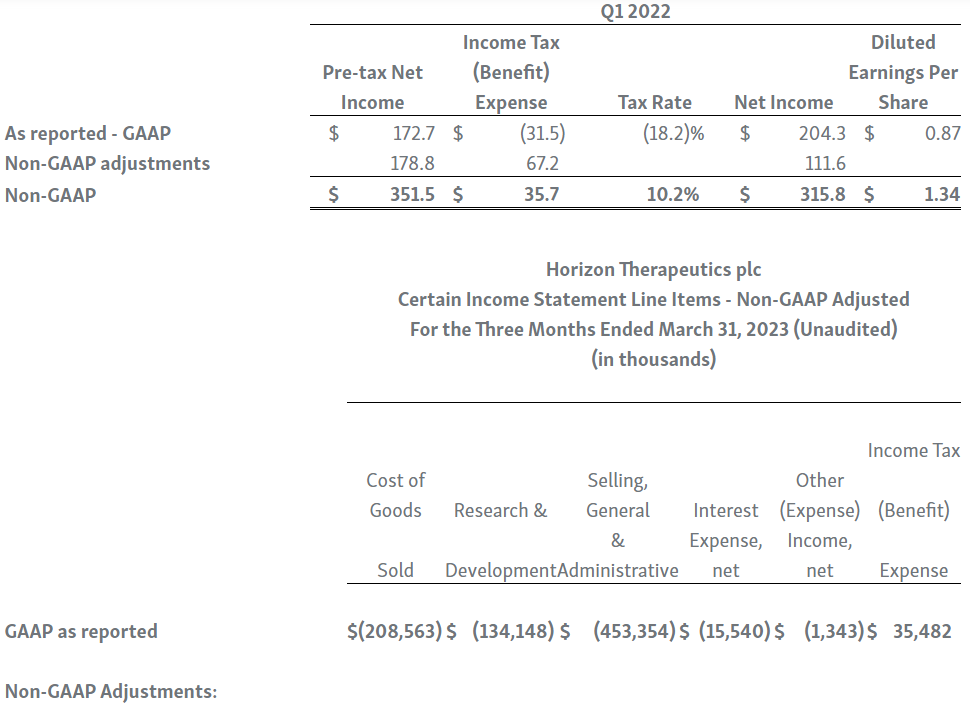
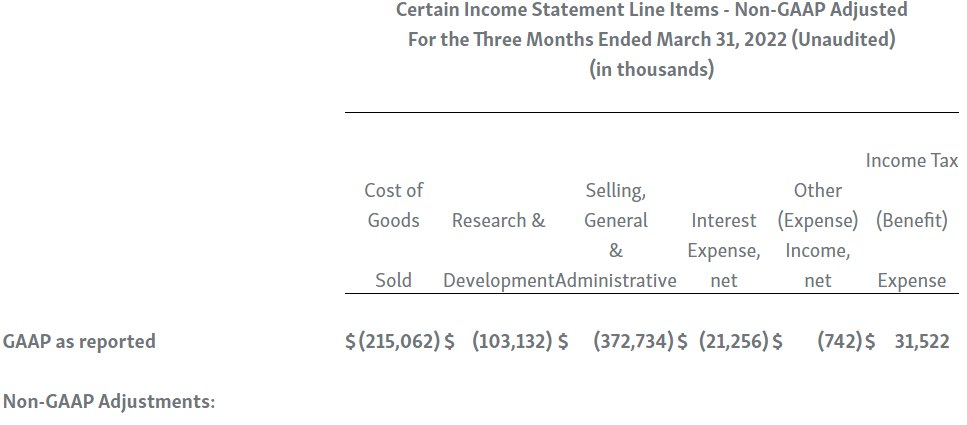
Horizon provides certain non-GAAP financial measures, including EBITDA, or earnings before interest, taxes, depreciation and amortization, adjusted EBITDA, non-GAAP net income, non-GAAP diluted earnings per share, non-GAAP gross profit and gross profit ratio, non-GAAP operating expenses, non-GAAP operating income, non-GAAP tax benefit (expense) and tax rate, non-GAAP operating cash flow and certain other non-GAAP income statement line items, each of which include adjustments to GAAP figures. These non-GAAP measures are intended to provide additional information on Horizon’s performance, operations, expenses, profitability and cash flows. Adjustments to Horizon’s GAAP figures exclude, as applicable, acquisition and/or divestiture-related costs, costs associated with our pending transaction with Amgen Inc., including responding to a second request review of the transaction by the United States Federal Trade Commission, manufacturing facility start-up costs, restructuring and realignment costs, as well as non-cash items such as share-based compensation, inventory step-up expense, depreciation and amortization, non-cash interest expense, goodwill and long-lived assets impairment charges, gain (loss) on equity security investments and other non-cash adjustments. Certain other special items or substantive events may also be included in the non-GAAP adjustments periodically when their magnitude is significant within the periods incurred. Horizon maintains an established non-GAAP cost policy that guides the determination of what costs will be excluded in non-GAAP measures. Horizon believes that these non-GAAP financial measures, when considered together with the GAAP figures, can enhance an overall understanding of Horizon’s financial and operating performance. The non-GAAP financial measures are included with the intent of providing investors with a more complete understanding of the Company’s historical and expected financial results and trends and to facilitate comparisons between periods and with respect to projected information. In addition, these non-GAAP financial measures are among the indicators Horizon’s management uses for planning and forecasting purposes and measuring the Company's performance. These non-GAAP financial measures should be considered in addition to, and not as a substitute for, or superior to, financial measures calculated in accordance with GAAP. The non-GAAP financial measures used by the Company may be calculated differently from, and therefore may not be comparable to, non-GAAP financial measures used by other companies.
Forward-Looking Statements
This press release contains forward-looking statements, including, but not limited to, statements related to the pending transaction with Amgen Inc., development, manufacturing and commercialization plans; expected timing of clinical trials; expected future milestones, pipeline expansions and regulatory approvals; potential market opportunities for, and benefits of, Horizon’s medicines and medicine candidates; expected impact of commercial strategies, clinical trial results and product label updates; and business and other statements that are not historical facts. These forward-looking statements are based on Horizon’s current expectations and inherently involve significant risks and uncertainties. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of these risks and uncertainties, which include, without limitation, whether the pending transaction with Amgen Inc. will be completed in a timely manner or at all; the parties’ ability to satisfy (or willingness to waive) the conditions to the consummation of the pending transaction with Amgen Inc., including with respect to required regulatory approvals; the effect of the pending transaction with Amgen Inc. on Horizon’s business relationships, operating results and business generally; risks that Horizon’s actual future financial and operating results may differ from its expectations or goals; Horizon’s ability to grow net sales from existing medicines; impacts of the COVID-19 pandemic and actions taken to slow its spread, including impacts on supplies and net sales of Horizon’s medicines and potential delays in clinical trials; impacts of the on-going war between Russia and Ukraine; changes in inflation, interest rates and general economic conditions; the availability of coverage and adequate reimbursement and pricing from government and third-party payers; Horizon’s ability to successfully implement its business strategies, including the risks that its medicine growth and global expansion initiatives and strategies may not be successful and that new challenges to growth may arise in the future; risks inherent in developing novel medicine candidates and existing medicines for new indications; whether additional clinical trial results or data analyses will be consistent with preliminary results, results from other trials or Horizon’s expectations; risks associated with regulatory approvals; risks in the ability to recruit, train and retain qualified personnel; competition, including generic competition; the ability to protect intellectual property and defend patents; regulatory obligations and oversight, including any changes in the legal and regulatory environment in which Horizon operates and those risks detailed from time-to-time under the caption "Risk Factors" and elsewhere in Horizon’s filings and reports with the SEC. Horizon undertakes no duty or obligation to update any forward-looking statements contained in this press release as a result of new information.



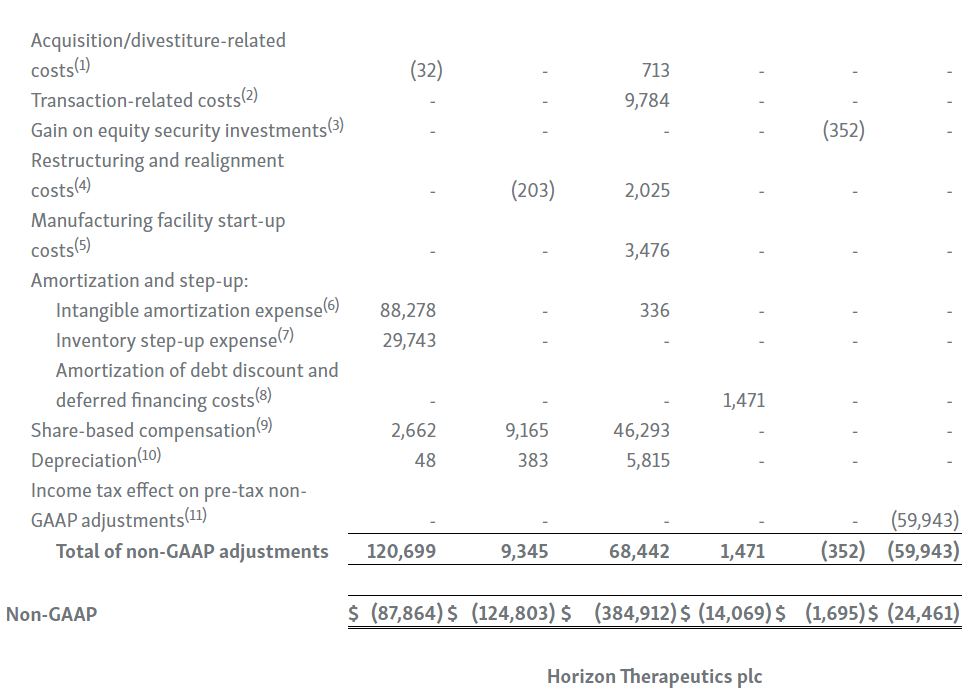
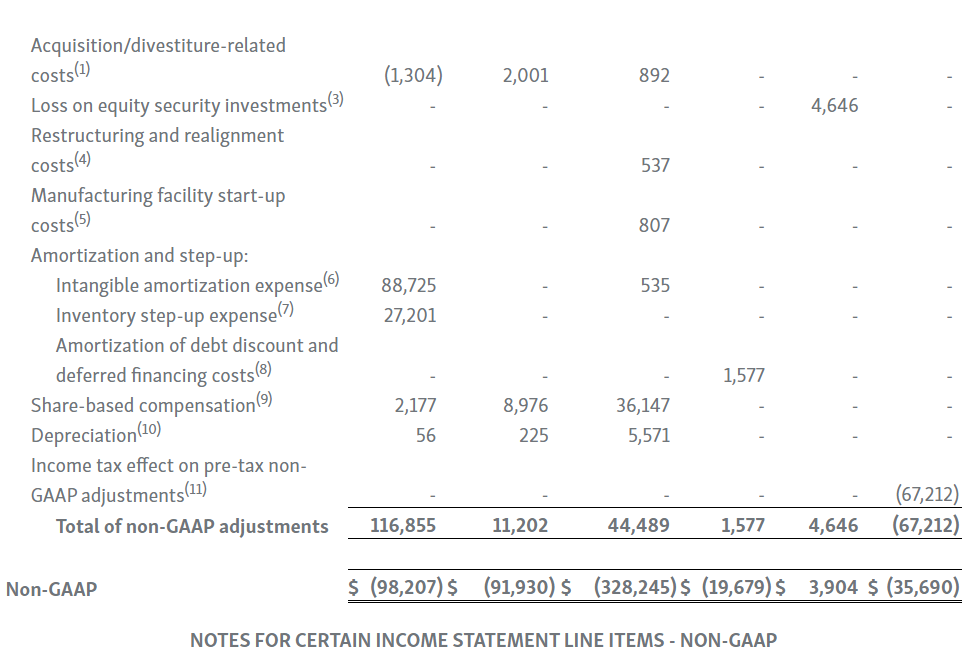
| 1. | Primarily represents transaction and integration costs, including, advisory, legal, consulting and certain employee-related costs, incurred in connection with our acquisitions and divestitures. |
| 2. | Primarily represents transaction-related costs, including, advisory, legal and consulting costs, incurred in connection with the Transaction with Amgen, including responding to the FTC’s second request. |
| 3. | We held investments in equity securities with readily determinable fair values of $7.4 million and $8.5 million as of March 31, 2023 and 2022, respectively, which are included in other long-term assets in the condensed consolidated balance sheet. For the three months ended March 31, 2023 and 2022, we recognized a net unrealized gain of $0.4 million and a net unrealized loss of $4.6 million, respectively, due to the change in fair value of these securities. |
| 4. | Primarily represents severance and consulting costs related to the wind down of our former inflammation business during 2022, and rent and maintenance charges as a result of vacating the leased Lake Forest office in the first quarter of 2021. |
| 5. | During the three months ended March 31, 2023 and 2022, we recorded $3.5 million and $0.8 million, respectively, of manufacturing facility start-up costs related to our drug product biologics manufacturing facility in Waterford, Ireland. |
| 6. | Intangible amortization expenses are primarily associated with our developed technology related to TEPEZZA, KRYSTEXXA, RAVICTI, UPLIZNA, PROCYSBI, ACTIMMUNE, RAYOS and BUPHENYL. |
| 7. | During the three months ended March 31, 2023 and 2022, we recognized $29.7 million and $27.2 million in cost of goods sold, respectively, for inventory step-up expense related to UPLIZNA inventory revalued in connection with the Viela Bio, Inc. acquisition. Because inventory step-up expense is related to an acquisition, will not continue indefinitely and has a significant effect on our gross profit, gross margin percentage and net income for all affected periods, we exclude inventory step-up expense from our non-GAAP financial measures. |
| 8. | Represents amortization of debt discount and deferred financing costs associated with our debt. |
| 9. | Represents share-based compensation expense associated with restricted stock unit and performance stock unit grants to our employees and non-employee directors, and our employee share purchase plan. |
| 10. | Represents depreciation expense related to our property, plant, equipment, software and leasehold improvements. |
| 11. | Income tax adjustments on pre-tax non-GAAP adjustments represent the estimated income tax impact of each pre-tax non-GAAP adjustment based on the statutory income tax rate of the applicable jurisdictions for each non-GAAP adjustment. |
Original source can be found here.