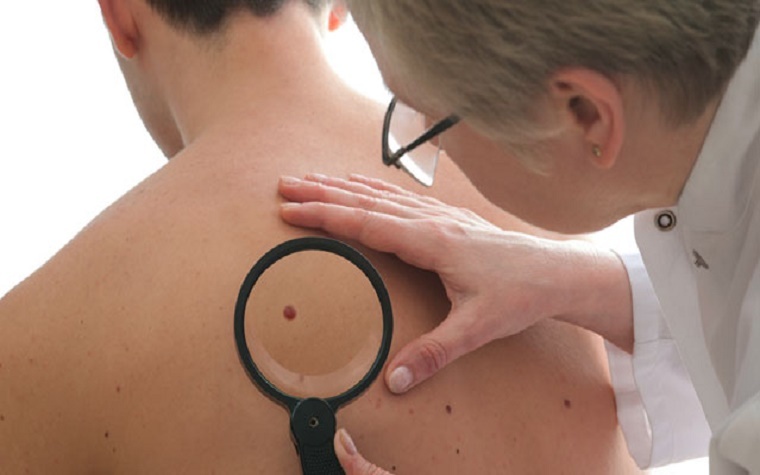
The U.S. Food and Drug Administration (FDA) said last week that Yervoy, which has been used since 2011 to treat melanoma, can now be used for Stage III melanoma patients after surgery to help decrease the likelihood that melanoma will recur.
Melanoma is the most aggressive form of skin cancer, causing an estimated 9,940 deaths in the U.S. each year. Melanoma is considered Stage III when it has spread to one or more lymph nodes.
"(The) approval of Yervoy extends its use to patients who are at high risk of developing recurrence of melanoma after surgery,” FDA Center for Drug Evaluation and Research Office of Hematology and Oncology Products Director Richard Pazdur said. “This new use of the drug in earlier stages of the disease builds on our understanding of the immune system’s interaction with cancer.”
Yervoy, a product of Bristol-Myers Squibb, is given intravenously and was initially approved for the treatment of late-stage, inoperable melanoma. It was the subject of a safety study on 951 patients.



